How Hard Water Effects a Swimming Pool
I. Understanding Water Hardness
Why is Water Hardness nearly as important as pH & Total Alkalinity?
After pH and Total Alkalinity, water hardness is another very important aspect of swimming pool water quality.
It is normally measured in parts per million (PPM) or miligrams per litre (mg/L) with a photometer or comparator. How hard or soft your water is means how much or how little calcium there is in your water.
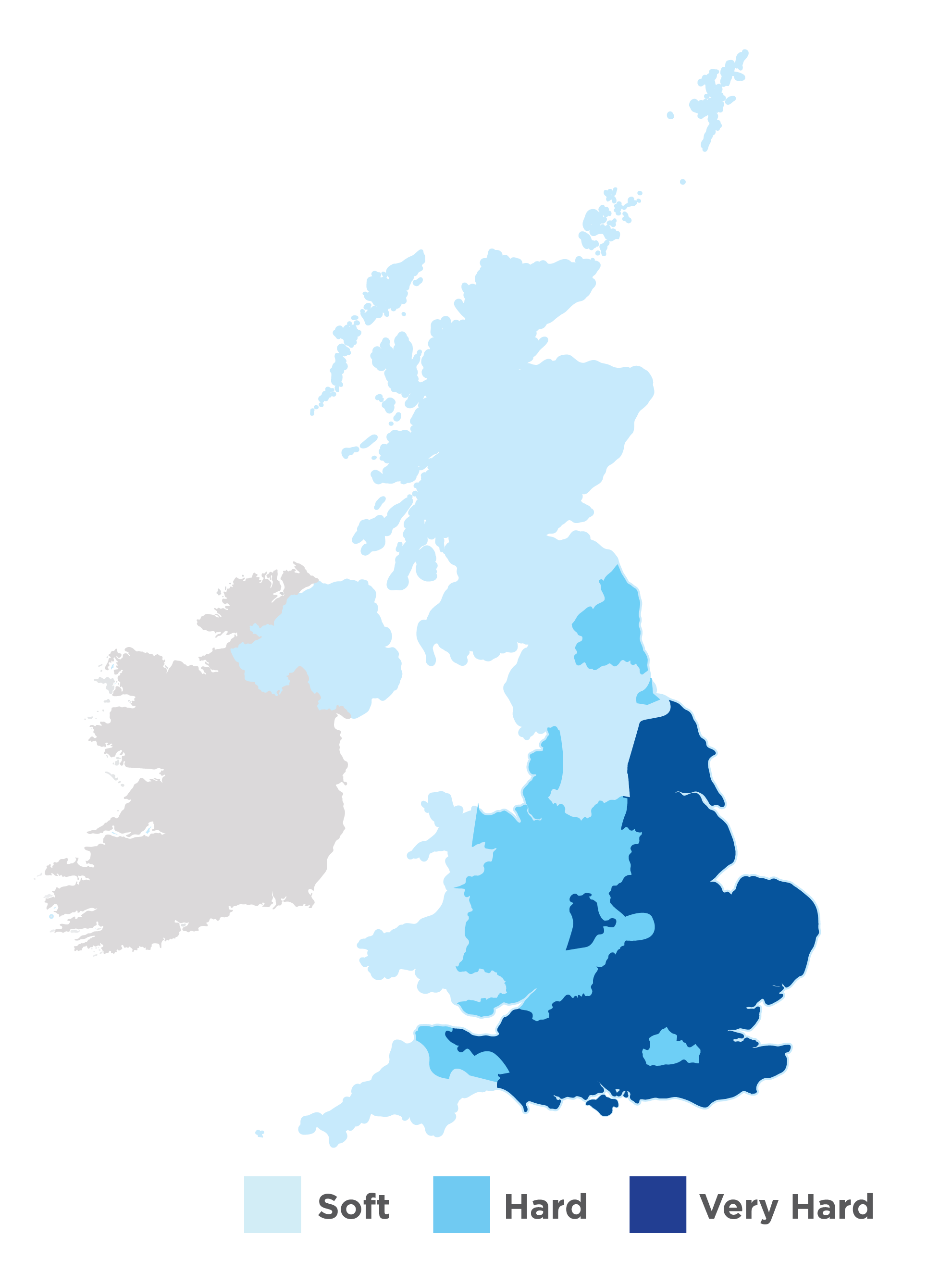
Source Water Hardness Across the UK:
When your pool is filled for the 1st time the water comes straight from your tap. You have no control over the calcium hardness of your source water and while it is completely adequate for drinking and cooking it is not always the best for your pool.
Across the UK the water hardness differs quite a lot - in Wales and Scotland the source water is very soft (and arguably the best water to make tea with) with a calcium content ranging between 0 to 50 mg/L (or PPM).
Compare that to the calcium hardness in source water in North London for example where the water is very hard with levels ranging between 200 and 350 mg/L (or PPM).
Where does it come from?
Calcium Carbonate is found in the acquifers of Limestone and Chalk where most of our water comes from.
We then purify and filter the water and deliver it straight to homes all over the UK. Depending on their source, the water will have a higher or lesser content of calcium carbonate concentrate in it – this directly relates to whether you have hard water (that forms calcium deposits in your kettle) or soft water (that has a tendency to corrode the element in the bottom of your kettle).
Hard water results in a calcium carbonate build up in heat exchangers, pumps , valves and water treatment cells that will eventually cause parts of the pool equipment to fail.
Soft water can often result in corrosion of any metallic elements in contact with the pool water.
II. Managing Water Hardness In Your Pool
What is the recommended level ?
PWTAG (The Pool Water Treatment Advisory Group who set the standards in the UK) recommend keeping your pool water hardness level between 80 and 200 mg/L (or PPM as CaCO3). The level at which your pool operates will depend on the source water and also on other chemicals that are added to the pool water, for example some disinfectants can help or hinder your control over the water hardness.
Does my choice of disinfectant have an effect on the water hardness in my pool?
Yes. There are different chemicals that can be used to disinfect your swimming pool water and these include:
- Sodium Hypochlorite (AKA Liquid Chlorine)
- Calcium Hypochlorite (AKA Chlorine Shock)
- Trichloroisocyanuric Acid (AKA Stabilised Chlorine)
For ease of use and to differentiate them the manufacturers call each of these three chemicals CHLORINE. All three are chlorine donors but have different characteristics and should be used in accordance with the manufacturers instructions.
As the name suggests – calcium hypochlorite – has calcium in it and so if you add this to your pool water you will be raising the calcium level as a consequence of disinfection. This is fine if you have very soft water because it will help to keep the calcium level at desirable levels. But if you have very hard water and find it hard to keep the hardness down then adding calcium hypochlorite to disinfect the pool will only lead to even higher levels in the pool. So the disinfectant should be chosen based on the source water.
Will hard water degrade my pool liner?
No. If the water is hard and your pH levels and Total Alkalinity levels are too high the soluability of the Calcium and other minerals becomes a lot lower and so this encourages their deposition on the bottom of the pool or on the pool walls. In a liner pool you can sweep this off with a brush but it is very important to get the levels under control in order to have clear, clean, sparkling water.
If you have deposits that are not pure white they may be the deposition of other minerals such as iron or copper and these can stain the liner.
Tiled Pools and Water Hardness:
Water with a low calcium concentration has a tendency to be corrosive and so in a tiled pool you may find that the grout is erroded over time. In a tiled pool it is important to keep the calcium levels at optimum because replacing the grout is difficult and costly.
How do I keep control over the Calcium Hardness of my pool water?
All the important levels in your pool - pH, Total Alkalinity, Calcium Hardness, Total Dissolved Solids - are all interrelated and so when one is out of control this will affect all the others. The most important one to start with is pH. If you can maintain the pH at the correct level (between 7.2 and 7.4) with no dramatic spikes or depressions then the others should fall into place.
However if you have a steady pH level and a steady Alkalinity level and your calcium hardness is too low then there are ways you can bring the calcium level up. This may be by using a calcium based disinfectant such as Calcium Hypochlorite.
If your Calcium levels are very high because of the source water being introduced then it is best to limit the amount of calcium added to the pool. So steer clear from Calcium based disinfectants and use Sodium Hypochlorite instead.
When using any pool chemicals always check the label and apply strictly within the manufacturers parameters. Never mix chemicals together.
